SMI's Quality Statement
We are committed to meeting customer and regulatory requirements through the effectiveness of our quality management system, high-quality products, superior service, and competitive prices.
ISO 13485 Medical Devices Certified
SMI has achieved the ISO 13485 Medical Devices Quality Management System certification, having proved our ability to provide silicone solutions for medical devices that consistently meet customer and regulatory requirements, through design, development, production, and distribution. SMI consistently achieves and exceeds standards to maintain certification upon each ISO audit. SMI is also a registered silicone contract manufacturer with the FDA.
SMI is a FDA Registered Contract Manufacturer
FDA Registration Number: 1831948
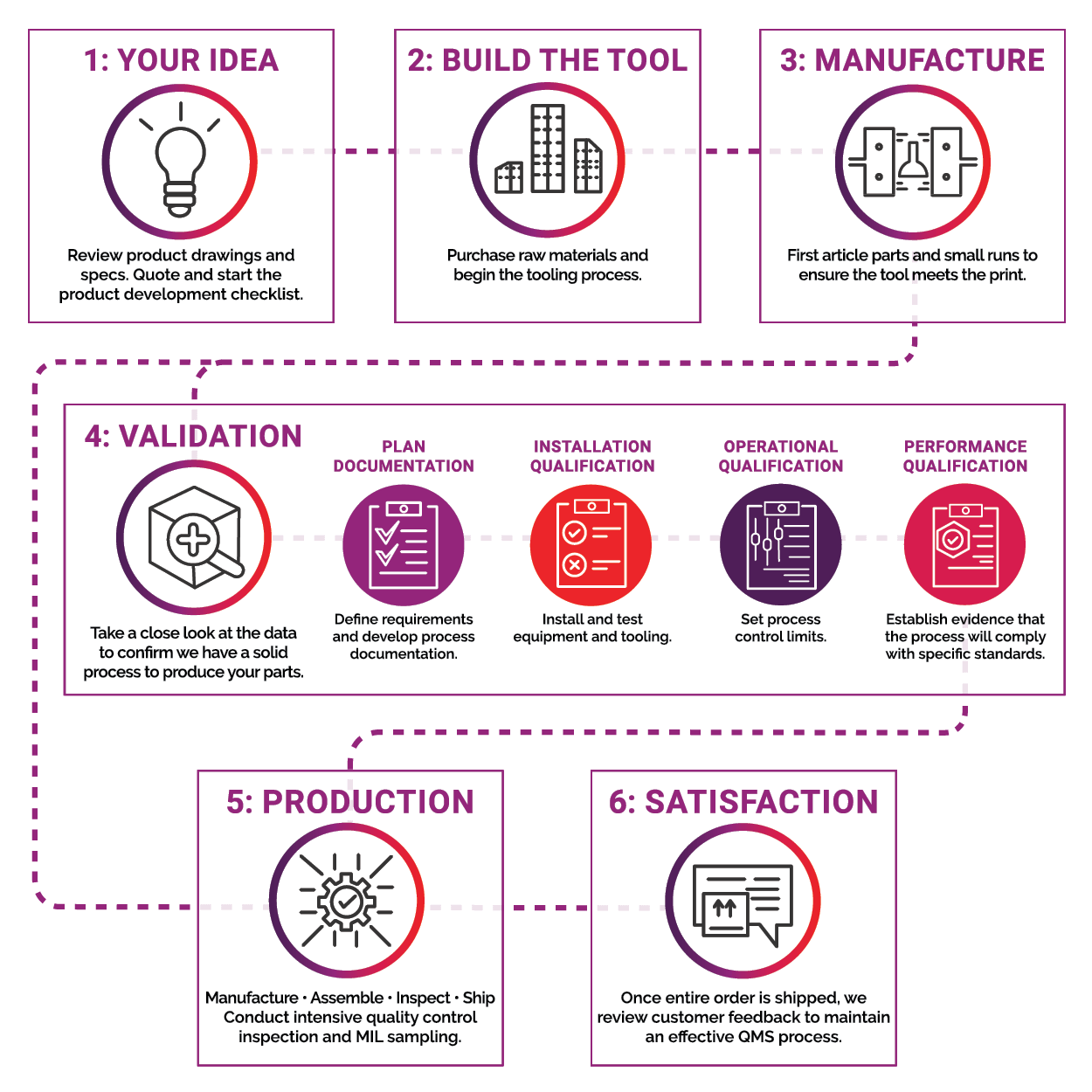
SMI's Process for Success
SMI experts have a proven process to ensure each product we engineer makes a big impact. It starts with your idea and ends with your satisfaction. To ensure a quality product, every engineered-product must pass SMI strict validation process prior to moving into production. Scroll down to view our process for success from start to finish.